1ЎўСЎФсМв ФЪТ»ЦЦЛбРФИЬТәЦРҙжФЪNO3ЁDЎўIЁDЎўClЁDЎўFe3+ЦРТ»ЦЦ»тјёЦЦАлЧУЈ¬ПтёГИЬТәЦРјУИлдеЛ®Ј¬деөҘЦКұ»»№ФӯЈ¬УЙҙЛНЖ¶ПИЬТәЦР
A.Т»¶Ёә¬УРIЁDЈ¬І»ДЬИ·¶ЁКЗ·сУРClЁD
B.Т»¶Ёә¬УРNO3ЁD
C.Т»¶ЁІ»ә¬УРFe3+ЎўClЁD
D.Т»¶Ёә¬УРIЁDЎўNO3ЁDәНClЎӘ
2ЎўСЎФсМв ПВБР·ҙУҰјИКЗСх»Ҝ»№Фӯ·ҙУҰУЦКЗАлЧУ·ҙУҰөДКЗЈЁ?Ј©
AЈ®УГCO»№ФӯСх»ҜМъАҙБ¶Мъ
BЈ®УГРҝәНПЎСОЛб·ҙУҰЦЖЗвЖш
CЈ®УГПЎБтЛбіэМъРв
DЈ®CO2НЁИліОЗеКҜ»ТЛ®ұд»лЧЗ
3ЎўСЎФсМв ДіОЮЙ«ИЬТәЦРә¬УРKЈ«ЎўClЈӯЎўOHЈӯЎўSO32ЎӘЎўSO42ЎӘЈ¬ОӘБЛјмСйіэOHЈӯНвөДЖдЛыЛщУРТхАлЧУЈ¬ДвУГСОЛбЎўПхЛбЎўПхЛбТшИЬТәЎўЗвСх»ҜұөИЬТәЎўдеЛ®әН·УМӘБщЦЦКФјБЈ¬ЙијЖИзПВКөСйІҪЦиЈ¬ІўјЗВјПа№ШПЦПуЎЈ
ПВБРУР№ШҪбВЫҙнОуөДКЗ(ЎЎЎЎ)

AЈ®КФјБўЫКЗAgNO3ИЬТәЈ¬КФјБўЭКЗHNO3Ј¬ПЦПу1ЦР°ЧЙ«іБөнКЗAgCl
BЈ®ПЦПу3ЦР°ЧЙ«іБөнКЗBaSO4
CЈ®ІъЙъПЦПу2өДАлЧУ·ҪіМКҪКЗBr2Ј«2H2OЈ«SO2=4HЈ«Ј«2BrЈӯЈ«SO42ЎӘ
DЈ®КФјБўЪКЗСОЛбЈ¬КФјБўЫКЗПхЛб
4ЎўСЎФсМв ФЪДіНёГчөДЗҝЛбРФИЬТәЦРЈ¬ДЬҙуБҝ№ІҙжөДОЮЙ«АлЧУЧйКЗ
A.K+ЎўNa+ЎўCu2+ЎўSO42-
B.Na+ЎўAl3+ЎўCl-ЎўNO3-
C.Na+ЎўK+ЎўCO32-ЎўCl-
D.K+ЎўSO42-ЎўCl-ЎўBa2+
5ЎўСЎФсМв Нщә¬IЈӯәНClЈӯөДПЎИЬТәЦРөОИлAgNO3ИЬТәЈ¬іБөнөДЦКБҝУлјУИлAgNO3ИЬТәМе»эөД№ШПөИзНјЛщКҫЎЈФтФӯИЬТәЦРc(IЈӯ)/c(ClЈӯ)өДұИЦөОӘ
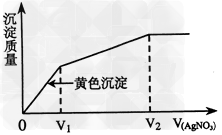
[? ]
AЈ®(V2ЈӯV1)/V1?
BЈ®V1/V2
CЈ®V1/(V2ЈӯV1Ј©?
DЈ®V2/V1