1ΓΔ―Γ‘ώΧβ œ¬Ν–±μ ω’ΐ»ΖΒΡ «(ΓΓΓΓ)
AΘ°ΦΉΆιΓΔ±ΫΓΔ““¥ΦΚΆ““œ©ΕΦΡήΖΔ…ζΦ”≥…Ζ¥”Π
BΘ°”ϊΫΪΒΑΑΉ÷ ¥”Υ°÷–Έω≥ωΕχ”÷≤ΜΗΡ±δΥϋΒΡ–‘÷ Θ§”ΠΦ”»κCuSO4»ή“Κ
CΘ°PPΥήΝœ(Ψέ±ϊœ©)ΫαΙΙΦρ Ϋ?CH2CH2CH2?
DΘ°ΕΓΆι¥ΏΜ·Ν―ΫβΩ…Α¥ΝΫ÷÷ΖΫ ΫΫχ––ΘΚC4H10®DΓζC2H4ΘΪC2H6ΘΜC4H10®DΓζCH4ΘΪC3H6
≤ΈΩΦ¥πΑΗΘΚD
±ΨΧβΫβΈωΘΚΦΉΆιΓΔ““¥Φ÷–≤Μ¥φ‘Ύ≤Μ±ΞΚΆΦϋΘ§≤ΜΡήΖΔ…ζΦ”≥…Ζ¥”ΠΘΜΦ”»κCuSO4»ή“Κ ΙΒΑΑΉ÷ ±δ–‘ΘΜΨέ±ϊœ©ΒΡΫαΙΙΦρ Ϋ « ΓΘΙ AΓΔBΓΔCœνΨυ¥μΓΘΕΓΆιΖ÷Ή”ΕœΝ¥ ±”–2÷÷ΖΫ ΫΘΚ
ΓΘΙ AΓΔBΓΔCœνΨυ¥μΓΘΕΓΆιΖ÷Ή”ΕœΝ¥ ±”–2÷÷ΖΫ ΫΘΚ
 Θ§Ζ÷±πΒΟΒΫC2H4ΚΆC2H6ΓΔCH4ΚΆC3H6ΓΘ
Θ§Ζ÷±πΒΟΒΫC2H4ΚΆC2H6ΓΔCH4ΚΆC3H6ΓΘ
±ΨΧβΡ―Ε»ΘΚ“ΜΑψ
2ΓΔ―Γ‘ώΧβ œ¬Ν–ΈΡΉ÷±μ ω”κΖ¥”ΠΖΫ≥Χ ΫΕ‘”Π«“’ΐ»ΖΒΡ «?Θ®?Θ©
AΘ° Β―ι “”Ο“ΚδεΚΆ±Ϋ‘Ύ¥ΏΜ·ΦΝΉς”Οœ¬÷Τδε±ΫΘΚ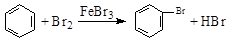
BΘ°δε““Άι÷–ΒΈ»κAgNO3»ή“ΚΦλ―ιΤδ÷–ΒΡδε‘ΣΥΊΘΚBrΘ≠+Ag+==AgBrΓΐ
CΘ°±ΫΖ”ΡΤ»ή“Κ÷–Ά®»κ…ΌΝΩCO2ΘΚCO2 + H2O + 2C6H5O- 2C6H5OH + CO32-
2C6H5OH + CO32-
DΘ° Β―ι “÷Τ““»≤ΘΚCaC2+H2O ΓζCaO+C2H2Γϋ
≤ΈΩΦ¥πΑΗΘΚA
±ΨΧβΫβΈωΘΚδε““ΆιΚΆœθΥα“χ «≤ΜΖ¥”ΠΒΡΘ§B≤Μ’ΐ»ΖΘΜ±ΫΖ”ΒΡΥα–‘«Ω”ΎΧΦΥα«βΡΤΒΡΘ§Υυ“‘C÷–ΒΡ…ζ≥…Έο «±ΫΖ”ΚΆΧΦΥα«βΡΤΘ§C≤Μ’ΐ»ΖΘΜΧΦΜ·ΗΤΚΆΥ°Ζ¥”Π…ζ≥…«β―θΜ·ΗΤΚΆ““»≤Θ§D≤Μ’ΐ»ΖΘ§¥πΑΗ―ΓAΓΘ
ΒψΤάΘΚΗΟΧβ «÷–Β»Ρ―Ε»ΒΡ ‘ΧβΘ§÷ς“Σ «ΩΦ≤ι―ß…ζΕ‘≥ΘΦϊ”–ΜζΖ¥”Π‘≠άμΒΡ λœΛΝΥΫβ≥ΧΕ»Θ§“β‘ΎΙ°ΙΧ―ß…ζΒΡΜυ¥Γ÷Σ ΕΘ§ΧαΗΏ―ß…ζΝιΜν‘Υ”ΟΜυ¥Γ÷Σ ΕΫβΨω ΒΦ Έ ΧβΒΡΡήΝΠΓΘ
±ΨΧβΡ―Ε»ΘΚ“ΜΑψ
3ΓΔΧνΩ’Χβ Θ®“ΜΘ©Ρ≥œ©Χΰ”κH2ΖΔ…ζΦ”≥…Ζ¥”ΠΚσΒΟΒΫΒΡ≤ζΈο «CH3ΓΣCH(CH3)2.
Δ≈‘≠ά¥œ©ΧΰΒΡΟϊ≥Τ «?
ΔΤ ‘≠ά¥œ©ΧΰΒΡΫαΙΙΦρ ΫΈΣΘΚ?
Δ« –¥≥ω‘≠ά¥ΒΡœ©Χΰ ΙδεΒΡΥΡ¬»Μ·ΧΦ»ή“ΚΆ …ΪΒΡΜ·―ßΖ¥”ΠΖΫ≥Χ ΫΘΚ
?
Θ®ΕΰΘ©Ρ≥ΆιΧΰΧΦΦήΫαΙΙ»γΆΦΥυ ΨΘΚ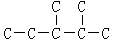 Θ§
Θ§
Θ®1Θ©¥ΥΆιΧΰΒΡ“Μδε¥ζΈο”–?÷÷
Θ®2Θ©»τ¥ΥΆιΧΰΈΣ»≤ΧΰΦ”«β÷ΤΒΟΘ§‘ρ¥Υ»≤ΧΰΒΡΫαΙΙΦρ ΫΈΣ?
Θ®3Θ©»τ¥ΥΆιΧΰΈΣΒΞœ©ΧΰΦ”«β÷ΤΒΟΘ§‘ρ¥Υœ©ΧΰΒΡΫαΙΙ”–?÷÷
≤ΈΩΦ¥πΑΗΘΚΘ®“ΜΘ©Θ®1Θ©2ΓΣΦΉΜυ±ϊœ©?Θ®2Θ©CH2=C(CH3)2.
(3) CH2=C(CH3)2 +Br2 CH2 BrΘ≠C Br(CH3)2
CH2 BrΘ≠C Br(CH3)2
Θ®ΕΰΘ©6ΘΜ  ?ΘΜ 5
?ΘΜ 5
±ΨΧβΫβΈωΘΚΘ®“ΜΘ©ΗΟΆιΧΰ «“λΕΓΆιΘ§‘ρ‘≠ά¥œ©Χΰ «2ΓΣΦΉΜυ±ϊœ©Θ§ΤδΫαΙΙΦρ Ϋ «CH2=C(CH3)2ΓΘœ©ΧΰΡήΚΆδεΥ°ΖΔ…ζΦ”≥…Ζ¥”ΠΘ§‘ρΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ Ϋ «CH2=C(CH3)2 +Br2 CH2 BrΘ≠C Br(CH3)2ΓΘ
CH2 BrΘ≠C Br(CH3)2ΓΘ
Θ®ΕΰΘ©Θ®1Θ©ΗυΨίΆιΧΰΒΡΫαΙΙΦρ ΫΩ…÷ΣΘ§Ζ÷Ή”÷–Κ§”–6άύ≤ΜΆ§ΒΡ«β‘≠Ή”Θ§Υυ“‘¥ΥΆιΧΰΒΡ“Μδε¥ζΈο”–6÷÷ΓΘ
Θ®2Θ©ΆιΧΰΖ÷Ή”÷–œύΝΎΒΡΧΦ‘≠Ή”÷Ν…ΌΗςΉ‘Κ§”–2Ηω“‘…œΒΡ«β‘≠Ή” ±Θ§≤≈ΡήΙΙ≥…ΧΦΧΦ»ΐΦϋΘ§Ψί¥ΥΩ…÷ΣΘ§ΗΟ»≤ΧΰΒΡΫαΙΙΦρ Ϋ « ΓΘ
ΓΘ
Θ®3Θ©Ά§―υΖ÷Ή”Ω…÷ΣΆιΧΰΖ÷Ή”÷–œύΝΎΒΡΧΦ‘≠Ή”÷Ν…ΌΗςΉ‘Κ§”–1Ηω“‘…œΒΡ«β‘≠Ή” ±Θ§≤≈ΡήΙΙ≥…ΧΦΧΦΥΪΦϋΘ§Ψί¥ΥΩ…÷ΣΘ§ΗΟœ©ΧΰΒΡΫαΙΙΦρ Ϋ”–5÷÷ΓΘ
ΒψΤάΘΚΗΟΧβ «ΗΏΩΦ÷–ΒΡ≥ΘΦϊΩΦΒψΚΆΧβ–ΆΘ§ τ”Ύ÷–Β»Ρ―Ε» ‘ΧβΒΡΩΦ≤ιΘ§ ‘ΧβΉέΚœ–‘«ΩΘ§‘ΎΉΔ÷ΊΕ‘―ß…ζΜυ¥Γ÷Σ ΕΙ°ΙΧΚΆ―ΒΝΖΒΡΆ§ ±Θ§≤ύ÷ΊΕ‘―ß…ζΡήΝΠΚΆΫβΧβΖΫΖ®ΒΡ÷ΗΒΦ”κ―ΒΝΖΘ§÷Φ‘ΎΩΦ≤ι―ß…ζΝιΜν‘Υ”ΟΜυ¥Γ÷Σ ΕΫβΨω ΒΦ Έ ΧβΒΡΡήΝΠΘ§”–άϊ”Ύ≈ύ―χ―ß…ζΒΡ¬ΏΦ≠ΆΤάμΡήΝΠΚΆΖΔ…ΔΥΦΈ§ΡήΝΠΓΘ
±ΨΧβΡ―Ε»ΘΚ“ΜΑψ
4ΓΔ―Γ‘ώΧβ ΧΰXΦ”H2ΚσΒΟΒΫΘ®CH3Θ©2CHCH2CH3Θ§‘ρXΩ…ΡήΈΣ
AΘ° 2ΓΣΦΉΜυΓΣ1ΓΣΕΓ»≤
BΘ° 3ΓΣΦΉΜυΓΣ1ΓΣΕΓ»≤
CΘ° 2ΓΣΦΉΜυΓΣ2ΓΣΕΓœ©
DΘ° 2ΓΣΦΉΜυΓΣ2ΓΣΕΓ»≤
≤ΈΩΦ¥πΑΗΘΚBC
±ΨΧβΫβΈωΘΚAD―Γœν÷–ΒΡΈο÷ ≤ΜΩ…Ρή¥φ‘ΎΘ§“άΨίΤδΟϊ≥Τ–¥≥ωΧΦΦήΩ…÷ΣΘ§≤ΩΖ÷ΧΦ‘≠Ή”“―Ψ≠≥§ΝΥΥΡΦέΘ§BC―Γœν÷–Έο÷ ΨυΖϊΚœΧβ“β
±ΨΧβΡ―Ε»ΘΚ“ΜΑψ
5ΓΔ―Γ‘ώΧβ ΟάΙζΩΒΡΥΕϊ¥σ―ßΒΡΈΚΩΦΩΥΥΙΘ®C.WilcoxΘ©ΥυΚœ≥…ΒΡ“Μ÷÷”–ΜζΖ÷Ή”Θ§ΨΆœώ“ΜΉπ Άε»Ρ≤ΡαΖπΘ§“ρΕχ≥ΤΈΣ Άε»Ρ≤ΡαΖ÷Ή”Θ®Υυ”–‘≠Ή”‘ΎΆ§“ΜΤΫΟφΘ©ΓΘ”–ΙΊΗΟ”–ΜζΈοΖ÷Ή”ΒΡΥΒΖ®≤Μ’ΐ»ΖΒΡ «
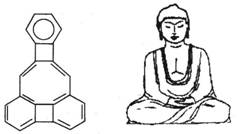
A.? τ”ΎΖΦœψΧΰ? B.? τ”Ύ±ΫΒΡΆ§œΒΈο
C.?Ζ÷Ή”÷–Κ§”–22ΗωΧΦ‘≠Ή”? D.?“Μ¬»¥ζΈο÷Μ”–3÷÷
≤ΈΩΦ¥πΑΗΘΚBD
±ΨΧβΫβΈωΘΚ¬‘
±ΨΧβΡ―Ε»ΘΚ“ΜΑψ