1ЎўСЎФсМв ФЪТ»ЦЦЛбРФИЬТәЦРҙжФЪNO3ЁDЎўIЁDЎўClЁDЎўFe3+ЦРТ»ЦЦ»тјёЦЦАлЧУЈ¬ПтёГИЬТәЦРјУИлдеЛ®Ј¬деөҘЦКұ»»№ФӯЈ¬УЙҙЛНЖ¶ПИЬТәЦР
A.Т»¶Ёә¬УРIЁDЈ¬І»ДЬИ·¶ЁКЗ·сУРClЁD
B.Т»¶Ёә¬УРNO3ЁD
C.Т»¶ЁІ»ә¬УРFe3+ЎўClЁD
D.Т»¶Ёә¬УРIЁDЎўNO3ЁDәНClЎӘ
2ЎўСЎФсМв ПВБРФЪИЬТәЦРҪшРРөДөзҪвЦКЦ®јдөД·ҙУҰТ»¶ЁДЬ№»·ўЙъөДКЗ
[? ]
ўЩУРЖшМеЙъіЙ
ўЪДЬ№»·ўЙъЦГ»»·ҙУҰ
ўЫЙъіЙДСИЬОпЦК
ўЬУРСОЙъіЙ
ўЭУРЛбЙъіЙ
AЈ®ўЩўЪўЬ
BЈ®ўЩўЫўЭ
CЈ®ўЩўЪўЫ
DЈ®ўЫўЬўЭ
3ЎўСЎФсМв ФЪЛ®ИЬТәЦРДЬБҝ№ІҙжөДТ»ЧйАлЧУКЗ
A.Na+ЎўBa2+ЎўCl-ЎўNO3-
B.Pb2+ЎўHg2+ЎўS2-ЎўSO42-
C.NH4+ЎўH+ЎўS2O32-ЎўPO43-
D.ClO-ЎўAl3+ЎўBr-ЎўCO32-
4ЎўМоҝХМв Ді»ҜС§СРҫҝРФС§П°РЎЧйМҪМЦFe3Ј«әН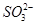 Ц®јд·ўЙъФхСщөД·ҙУҰЈ¬ЗлДгТ»ЖрІОУлІўРӯЦъЛыГЗНкіЙКөСйЎЈ(1)МбіцІВПлЈә
Ц®јд·ўЙъФхСщөД·ҙУҰЈ¬ЗлДгТ»ЖрІОУлІўРӯЦъЛыГЗНкіЙКөСйЎЈ(1)МбіцІВПлЈә
јЧН¬С§ИПОӘ·ўЙъСх»Ҝ»№Фӯ·ҙУҰЈ¬Жд·ҙУҰ·ҪіМКҪОӘ____________________________________ЎЈ
ТТН¬С§ИПОӘ·ўЙъЛ«Л®Ҫв·ҙУҰЈ¬Жд·ҙУҰ·ҪіМКҪОӘ2Fe3Ј«Ј«3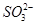 Ј«6H2O===2Fe(OH)3(ҪәМе)Ј«3H2SO3ЎЈ
Ј«6H2O===2Fe(OH)3(ҪәМе)Ј«3H2SO3ЎЈ
ЎӯЎӯ
(2)КөСйСйЦӨЈә
ұыН¬С§ЙијЖБЛПВБРКөСйАҙМҪҫҝ·ҙУҰөДҝЙДЬРФЎЈ
ўЩОӘБЛјмСйЛщУГNa2SO3КЗ·сұдЦКЈ¬УҰСЎУГөДКФјБКЗ___?_____ЎЈ
ўЪИЎ5 mL FeCl3ИЬТәУЪКФ№ЬЦРЈ¬ЦрөОјУИлNa2SO3ИЬТәЦБ№эБҝЈ¬№ЫІмөҪИЬТәСХЙ«УЙ»ЖЙ«ұдОӘәмЧШЙ«(ОЮЖшЕЭІъЙъЈ¬ТІОЮіБөнЙъіЙ)ЎЈ
ўЫҪ«ўЪИЬТә·ЦіЙБҪөИ·ЭЈ¬ЖдЦРТ»·ЭјУИлПЎСОЛбЦБ№эБҝЈ¬ФЩјУИлBaCl2ПЎИЬТәЈ¬УР°ЧЙ«іБөнЙъіЙЈ»БнТ»·ЭөОИлјёөОKSCNИЬТәЈ¬ИЬТәұдіЙСӘәмЙ«ЎЈ
(3)өГіцҪбВЫЈә
ўЩёщҫЭұыН¬С§өДКөСйөГіцөДҪбВЫКЗЈә________________?____________ЎЈ
ўЪКөСйўЫЦРИЬТәұдіЙСӘәмЙ«өДУР№ШАлЧУ·ҪіМКҪОӘ_________?____ЎЈ
(4)НШХ№МҪҫҝЈә
ўЩ¶ЎН¬С§ФЪFeCl3ИЬТәЦРјУИлNa2CO3ИЬТәЈ¬№ЫІмөҪәмәЦЙ«іБөнІўЗТІъЙъОЮЙ«ЖшМеЈ¬ёГ·ҙУҰөДАлЧУ·ҪіМКҪКЗ________?__________________ЎЈ
ўЪҙУРОКҪЙПҝҙЈ¬Na2CO3әНNa2SO3ПаЛЖЈ¬ө«КЗҙУЙПКцКөСйЦРҝЙТФҝҙөҪЈ¬БҪХЯөДЛ®ИЬТәУлВИ»ҜМъИЬТә·ҙУҰөДПЦПуІоұрәЬҙуЈ¬ЖдҝЙДЬөДФӯТтіэ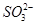 Л®ҪвДЬБҰҪП
Л®ҪвДЬБҰҪП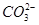 РЎНвЈ¬»№УР_?_______ЎЈ
РЎНвЈ¬»№УР_?_______ЎЈ
5ЎўСЎФсМв ПВБРКфУЪөзҪвЦКІўДЬөјөзөДОпЦККЗ
A.өн·Ы
B.KNO3ИЬТә
C.FeCl3ҫ§Ме
D.ИЫИЪөДNa2O