1ЎўСЎФсМв Птә¬УРПВБРАлЧУөДИЬТәЦРЈ¬НЁИлККБҝөДВИЖш»бТэЖрАлЧУЕЁ¶ИјхЙЩөДКЗЈЁЎЎЎЎЈ©
ўЩHCO3-ЎЎўЪSO32-ўЫOH-ЎЎўЬFe2+Ј®
AЈ®ўЩўЪ
BЈ®ўЫўЬ
CЈ®ўЩўЪўЫ
DЈ®ўЩўЪўЫўЬ
ІОҝјҙр°ёЈәВИЖшНЁИлИЬТәЦРЈ¬УлЛ®·ҙУҰ·ўЙъCl2+H2OЁTHCl+HClOЈ¬
ФтўЩСОЛбУлHCO3-·ҙУҰЙъіЙЛ®әН¶юСх»ҜМјЈ¬ТэЖрАлЧУЕЁ¶ИјхЙЩЈ»
ўЪСОЛбУлSO32-·ҙУҰЙъіЙЛ®әН¶юСх»ҜБтЈ¬ТэЖрАлЧУЕЁ¶ИјхЙЩЈ»
ўЫСОЛбУлOH-·ҙУҰЙъіЙЛ®Ј¬ТэЖрАлЧУЕЁ¶ИјхЙЩЈ»
ўЬҙОВИЛбУлFe2+·ўЙъСх»Ҝ»№Фӯ·ҙУҰЈ¬ТэЖрАлЧУЕЁ¶ИјхЙЩЈ»
№КСЎDЈ®
ұҫМвҪвОцЈә
ұҫМвДС¶ИЈәТ»°г
2ЎўСЎФсМв ПВБР»ҜәПОпІ»ДЬНЁ№эөҘЦКЦұҪУ»ҜәП¶шЦЖөГөДКЗЈЁЎЎЎЎЈ©
AЈ®CuCl2
BЈ®AlCl3
CЈ®FeCl2
DЈ®NaCl
ІОҝјҙр°ёЈәAЎўөҘЦКНӯәНВИЖш»ҜәПЙъіЙCuCl2Ј¬№КAІ»·ыәПЈ»
BЎўВБәНВИЖш·ҙУҰЙъіЙAlCl3Ј¬№КBІ»·ыәПЈ»
CЎўМъәНВИЖш·ҙУҰЙъіЙFeCl3Ј¬№КC·ыәПЈ»
DЎўДЖәНВИЖшЙъіЙNaClЈ¬№КDІ»·ыәПЈ»
№КСЎЈәCЈ®
ұҫМвҪвОцЈә
ұҫМвДС¶ИЈәТ»°г
3ЎўМоҝХМв ЈЁ7·ЦЈ©ВИЛ®ЦРҙжФЪөД·ЦЧУУРЈә?әНH2OЈ¬АлЧУУРЈә?әНј«ОўБҝөДOHЈӯЈ¬ПтЖдЦРөОјУјёөОКҜИпЈ¬ИЬТәПИұдәмЈ¬әуНКЙ«өДФӯТтКЗЈә???
ІОҝјҙр°ёЈәCl2ЎўHClO?ЈЁ2·ЦЈ©? H+ ? ClЈӯ ClOЈӯЈЁ3·ЦЈ©?ВИЛ®ЦРөДH+
? ClЈӯ ClOЈӯЈЁ3·ЦЈ©?ВИЛ®ЦРөДH+ К№КҜИпПФәмЙ«Ј¬әуHClOөДЗҝСх»ҜРФУЦК№әмЙ«НКИҘЎЈЈЁ2·ЦЈ©
К№КҜИпПФәмЙ«Ј¬әуHClOөДЗҝСх»ҜРФУЦК№әмЙ«НКИҘЎЈЈЁ2·ЦЈ©
ұҫМвҪвОцЈәВФ
ұҫМвДС¶ИЈәТ»°г
4ЎўјтҙрМв УГИзНјЛщКҫЧ°ЦГҝЙТФНкіЙТ»ПөБРКөСйЈЁНјЦРјРіЦЧ°ЦГТСВФИҘЈ©
Зл»ШҙрПВБРОКМвЈә
ўсИфЖшМеaКЗCl2Ј¬Ч°ЦГAЎўBЎўCЦРөДКФјБТАҙООӘЈәFeCl2ИЬТәЎўөн·ЫKIИЬТәЎўКҜИпИЬТәЈ®
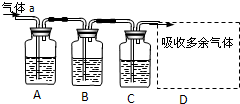
| Ч°ЦГ | A | B | C | D
КФјБ
FeCl2ИЬТә
өн·ЫKIИЬТә
КҜИпИЬТә
______
ПЦПу
ИЬТәУЙЗіВМЙ«ұдОӘЧШ»ЖЙ«
______
______
АлЧУ·ҪіМКҪ
______
______
______
|
IIИфЖшМеaКЗSO2Ј¬Ч°ЦГAЎўBЎўCЦРөДКФјБТАҙООӘЈәЖ·әмИЬТәЎўЛбРФёЯГМЛбјШИЬТәЎўЗвБтЛбЈЁБт»ҜЗвөДЛ®ИЬТәЈ©Ј®
| Ч°ЦГ | A | B | C
КФјБ
Ж·әмИЬТә
ЛбРФёЯГМЛбјШИЬТә
Бт»ҜЗвөДЛ®ИЬТә
ПЦПу
______
ЧПЙ«НКИҘ
______
МеПЦSO2өДРФЦК
______
______
Сх»ҜРФ
|
ЈЁ1Ј©РҙіцCЦР·ўЙъ·ҙУҰөД»ҜС§·ҪіМКҪ______Ј®
ЈЁ2Ј©DЧ°ЦГЦРУГЗвСх»ҜДЖИЬТәАҙОьКХSO2Ј¬ОӘБЛ·АЦ№ө№ОьЈ¬УҰёГСЎУГПВБРЧ°ЦГЦРөД______ЈЁМоРтәЕЈ©Ј®
ІОҝјҙр°ёЈәўсИфЖшМеaКЗCl2Ј¬Ч°ЦГAЎўBЎўCЦРөДКФјБТАҙООӘЈәFeCl2ИЬТәЎўөн·ЫKIИЬТәЎўКҜИпИЬТәЈ¬
ВИЖшУлAЦРВИ»ҜСЗМъ·ҙУҰЙъіЙВИ»ҜМъЈ¬№ЫІмөҪИЬТәУЙЗіВМЙ«ұдОӘ»ЖЙ«Ј¬·ўЙъөДАлЧУ·ҙУҰОӘCl2+2Fe2+=2Fe3++2Cl-Ј¬
УлBЦРөн·ЫKI·ҙУҰЙъіЙөвөҘЦКЈ¬№ЫІмөҪИЬТәұдА¶Ј¬·ўЙъөДАлЧУ·ҙУҰОӘCl2+2I-=I2+2Cl-Ј¬
УлCЦРЧПЙ«КҜИпКФТәПИұдәмәуНКЙ«Ј¬DОӘОІЖшҙҰАнЧ°ЦГЈ¬УлDЦРNaOH·ҙУҰЙъіЙNaClЎўNaClOәНЛ®Ј¬АлЧУ·ҙУҰОӘCl2+2OH-=Cl-+ClO-+2H2OЈ¬№Кҙр°ёОӘЈәЧ°ЦГABCDКФјБNaOHИЬТәПЦПуИЬТәұдА¶ПИұдәмәуНКЙ«АлЧУ·ҪіМКҪCl2+2Fe2+=2Fe3++2Cl-Cl2+2I-=I2+2Cl-Cl2+2OH-=Cl-+ClO-+2H2OIIИфЖшМеaКЗSO2Ј¬Ч°ЦГAЎўBЎўCЦРөДКФјБТАҙООӘЈәЖ·әмИЬТәЎўЛбРФёЯГМЛбјШИЬТәЎўЗвБтЛбЈЁБт»ҜЗвөДЛ®ИЬТәЈ©Ј¬
¶юСх»ҜБтК№AЦРЖ·әмНКЙ«Ј¬МеПЦ¶юСх»ҜБтҫЯУРЖҜ°ЧРФЈ¬УлBЦРёЯГМЛбјШ·ўЙъСх»Ҝ»№Фӯ·ҙУҰЈ¬МеПЦЖд»№ФӯРФЈ¬УлCЦРБт»ҜЗвөДЛ®ИЬТә·ўЙъСх»Ҝ»№Фӯ·ҙУҰЙъіЙБтәНЛ®Ј¬№ЫІмөҪІъЙъөӯ»ЖЙ«іБөнЈ¬МеПЦЖдСх»ҜРФЈ¬№Кҙр°ёОӘЈә
Ч°ЦГABCКФјБПЦПуәмЙ«НКИҘЙъіЙөӯ»ЖЙ«іБөнМеПЦSO2өДРФЦКЖҜ°ЧРФ»№ФӯРФЈЁ1Ј©CЦР¶юСх»ҜБтУлБт»ҜЗв·ҙУҰЙъіЙБтәНЛ®Ј¬ёГ·ҙУҰОӘSO2+2H2SЁT3SЎэ+2H2OЈ¬№Кҙр°ёОӘЈәSO2+2H2SЁT3SЎэ+2H2OЈ»
ЈЁ2Ј©ЛщёшөДЧ°ЦГІ»»біцПЦДЪНвС№ЗҝІоЈ¬ҫНДЬ·АЦ№ө№ОьЈ¬НјЦРўЩўЫҫЯУР»әіеөДЗтРОҪб№№Ј¬І»»бРОіЙС№ЗҝІоЈ¬ФтДЬЖр·Аө№ОьөДЧчУГЈ¬№Кҙр°ёОӘЈәўЩўЫЈ®
ұҫМвҪвОцЈә
ұҫМвДС¶ИЈәТ»°г
5ЎўМоҝХМв ўсЈ®ИзНјЛщКҫAЎ«HОӘКөСйКТіЈјыөДТЗЖчЎўЧ°ЦГЈЁІҝ·Ц№М¶ЁјРіЦЧ°ЦГВФИҘЈ©Ј¬ЗлёщҫЭТӘЗу»ШҙрПВБРОКМвЈә
ЈЁ1Ј©AТЗЖчөДГыіЖКЗ______Ј¬іЈУГУЪ______Ј®
ЈЁ2Ј©УГНјЦРЛщКҫТЗЖчЙијЖТ»МЧЦЖИЎТ»ЖҝёЙФпЎўҙҝҫ»°ұЖшөДКөСйЧ°ЦГЈ¬ТЗЖчөДБ¬ҪУЛіРтКЗЈЁУГ№ЬҝЪЧЦДё·ыәЕұнКҫЈ©ЈәjҪУ______Ј®
ўтЈ®ОӘБЛСРҫҝCl2өДРФЦКЈ¬ЙијЖИзНјЛщКҫЧ°ЦГҪшРРКөСйЈЁЧ°ЦГўуЦРјРіЦЧ°ЦГТСВФИҘЈ¬ЖдЦРaОӘёЙФпөДЖ·әмКФЦҪЈ¬bОӘКӘИуөДЖ·әмКФЦҪЈ©Ј®
ЈЁ3Ј©јУИлТ©Ж·З°Ј¬јмІйўсЦРЖшМе·ўЙъЧ°ЦГЖшГЬРФөДІЩЧчКЗ______Ј®
ЈЁ4Ј©КөСй№эіМЦРЈ¬Ч°ЦГўфЦРөДКөСйПЦПуОӘ______Ј®
ЈЁ5Ј©КөСйҪбКшәуЈ¬ёГЧйН¬С§ФЪЧ°ЦГўуЦР№ЫІмөҪbөДәмЙ«НКИҘЈ¬ө«КЗІўОҙ№ЫІмөҪЎ°aОЮГчПФұд»ҜЎ°ХвТ»ФӨЖЪПЦПуЈ®ОӘБЛҙпөҪХвТ»КөСйДҝөДЈ¬ДгИПОӘУҰФЪ______Ц®јдЈЁСЎМоЧ°ЦГРтәЕЈ©»№РиМнјУТ»ёцПҙЖшЖҝЈ¬ёГЧ°ЦГөДЧчУГКЗ______Ј®
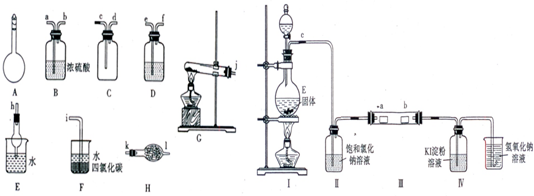
ІОҝјҙр°ёЈәЈЁ1Ј©УЙНјҝЙЦӘAОӘИЭБҝЖҝЈ¬ИЭБҝЖҝҝЙУГАҙЕдЦЖТ»¶ЁОпЦКөДБҝЕЁ¶ИөДИЬТәЈ¬
№Кҙр°ёОӘЈәИЭБҝЖҝЈ»ЕдЦЖТ»¶ЁОпЦКөДБҝЕЁ¶ИөДИЬТәЈ»
ЈЁ2Ј©ЦЖИЎТ»ЖҝёЙФпЎўҙҝҫ»°ұЖшЈ¬ФтАыУГ№ММеУл№ММејУИИЧ°ЦГЎўёЙФпЧ°ЦГЈ¬ЕЕҝХЖш·ЁКХјҜЧ°ЦГЎўОІЖшҙҰАнЧ°ЦГЈ¬GОӘјУИИЧ°ЦГЈ¬HОӘёЙФпЧ°ЦГЈ¬ҙуҝЪҪшЈ¬РЎҝЪіцЈ¬
CОӘКХјҜЧ°ЦГЈ¬°ұЖшөДГЬ¶ИiҝХЖшөДРЎЈ¬Фт¶МҪшіӨіцЈ¬E»тF¶јҝЙУГАҙҙҰАнОІЖшЈ¬јҙБ¬ҪУЛіРтОӘjҪУkЎўlҪУdЎўcҪУh»тIЈ¬№Кҙр°ёОӘЈәҪУkЎўlҪУdЎўcҪУh»тIЈ»
ЈЁ3Ј©Ч°ЦГўсЦРУР·ЦТәВ©¶·Ј¬АыУГТәІо·ЁАҙјмІйЧ°ЦГөДЖшГЬРФЈ¬ІЩЧчОӘУГЦ№Л®јРјРЧЎCҙҰПрЖӨ№ЬЈ¬И»әуПт·ЦТәВ©¶·ЦРјУЛ®Ј¬ҙтҝӘ·ЦТәВ©¶·»оИыЈ¬УРТәМеТәГжёЯУЪЙХЖҝТәГжЈ¬ө«І»БчПВЈ¬ФтЖшГЬРФәГЈ¬
№Кҙр°ёОӘЈәУГЦ№Л®јРјРЧЎCҙҰПрЖӨ№ЬЈ¬И»әуПт·ЦТәВ©¶·ЦРјУЛ®Ј¬ҙтҝӘ·ЦТәВ©¶·»оИыЈ¬УРТәМеТәГжёЯУЪЙХЖҝТәГжЈ¬ө«І»БчПВЈ¬ФтЖшГЬРФәГЈ»
ЈЁ4Ј©ТтВИЖшДЬҪ«өвАлЧУСх»ҜОӘөвөҘЦКЈ¬өвУцөн·ЫұдА¶Ј¬Фт»б№ЫІмөҪОЮЙ«ТәМеұдА¶Й«Ј¬№Кҙр°ёОӘЈәОЮЙ«ТәМеұдА¶Й«Ј»
ЈЁ5Ј©ТтaОӘёЙФпөДЖ·әмКФЦҪЈ¬bОӘКӘИуөДЖ·әмКФЦҪЈ¬Иф№ЫІмөҪaОЮГчПФұд»ҜЈ¬bөДәмЙ«НКИҘЈ¬ФтРиТӘФЪўтЎўўуЦ®јдјУТ»ёцПҙЖшЖҝАҙёЙФпВИЖшЈ¬№Кҙр°ёОӘЈәўтЎўўуЈ»ёЙФпВИЖшЈ®
ұҫМвҪвОцЈә
ұҫМвДС¶ИЈәјтөҘ