1ЎўСЎФсМв ПтЛб»Ҝ№эөДMnSO4ИЬТәЦРөОјУ(NH4)2S2O8(№э¶юБтЛбп§)ИЬТә»б·ўЙъ·ҙУҰЈәMn2Ј«Ј«S2O82-Ј«H2OЁDЎъMnO4-Ј«HЈ«Ј«SO42-ЎЈПВБРЛө·ЁІ»ХэИ·өДКЗ(ЎЎЎЎ)
AЈ®ҝЙТФАыУГёГ·ҙУҰјмСйMn2Ј«
BЈ®Сх»ҜРФұИҪПЈәS2O82-ЈҫMnO4-
CЈ®MnSO4ИЬТәҝЙТФК№УГСОЛбЛб»Ҝ
DЈ®ИфУР0.1 molСх»ҜІъОпЙъіЙЈ¬ФтЧӘТЖөзЧУ0.5 mol
ІОҝјҙр°ёЈәC
ұҫМвҪвОцЈәУЙУЪS2O82-өДСх»ҜРФҙуУЪMnO4-өДЈ¬өұУГСОЛбЛб»ҜКұClЈӯ»бұ»S2O82-Сх»ҜОӘCl2ЎЈ
ұҫМвДС¶ИЈәТ»°г
2ЎўКөСйМв ЈЁ15·ЦЈ©ОӘБЛМҪҫҝH2O2ЎўH2SO3әНBr2Сх»ҜРФөДПа¶ФЗҝИхЈ¬ЙијЖИзПВКөСйЈЁјРіЦТЗЖчТСВФИҘЈ©ЎЈЗл»ШҙрПВБРОКМвЈә
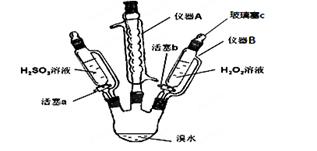
ЈЁ1Ј©ТЗЖчAөДГыіЖ_________Ј¬ЖдЧчУГКЗ___________ЎЈ
ЈЁ2Ј©УГТЗЖчBөОјУТәМеІўІ»РиТӘҙтҝӘІЈБ§ИыcЈ¬ФӯТтКЗ____________________________ЎЈ
ЈЁ3Ј©КөСйјЗВјИзПВЈЁЗлІ№И«ҝХ°ЧЈ©Јә
ІҪЦи
| КөСйІЩЧч
| КөСйПЦПу
| КөСйҪбВЫ
|
ўс
| ҙтҝӘ»оИыaЈ¬ЦрөОјУИлH2SO3ИЬТәЦБ№эБҝ
| ________________
| __________________________
|
ўт
| ПтІҪЦиўсЛщөГИЬТәЦРЦрөОјУИлH2O2ИЬТә
| ёХҝӘКјИЬТәСХЙ«ОЮГчПФұд»ҜЈ¬јМРшөОјУЈ¬ИЬТәұдОӘіИ»ЖЙ«
| __________________________
|
ЈЁ4Ј©ІҪЦиўтЦРЈ¬ҝӘКјКұСХЙ«ОЮГчПФұд»ҜөДФӯТтКЗЈЁРҙіцТ»МхЈ©_______________________Ј¬
ІҪЦиўсЦР·ҙУҰөДАлЧУ·ҪіМКҪ_________________________________________________Ј¬
ІҪЦиўтЦРЦчТӘ·ҙУҰөДАлЧУ·ҪіМКҪ_____________________________________________ЎЈ
ІОҝјҙр°ёЈәЈЁ1Ј©ЗтРОАдДэ№ЬЈЁ»тАдДэ№ЬЈ© »ШБчдеЈЁ»тТФГвде»У·ўөИЈ© ЈЁ2Ј©өОТәВ©¶·әНИэҫұЖҝЖшС№ПаНЁ
ЈЁ3Ј©ўсЈәіИ»ЖЙ«НКИҘ Br2өДСх»ҜРФҙуУЪH2SO3 ўтЈәH2O2өДСх»ҜРФҙуУЪBr2
ЈЁ4Ј©ІҪЦи1өДH2SO3УР№эБҝЈ¬H2O2ПИәНH2SO3·ҙУҰЈЁH2O2ЕЁ¶ИРЎ»тBrЈӯУлH2O2·ҙУҰВэөИ¶јҝЙЈ©
H2SO3+Br2+H2O=4H++SO42-+2Br- H2O2+2Br-Ј«2H+ЈҪBr2Ј«2H2O
ұҫМвҪвОцЈә
КФМвҪвОцЈәЈЁ1Ј©ТЗЖчAөДГыіЖКЗЗтРОАдДг№ЬЈ¬ЖдЧчУГКЗК№деХфЖыАдДэ»ШБчЎЈЈЁ2Ј©УГТЗЖчBөОјУТәМеІўІ»РиТӘҙтҝӘІЈБ§ИыcЈ¬ФӯТтКЗёГТЗЖчУлИэҫұЖҝПаН¬Ј¬ЖшС№ПаөИЈ¬ТәМеДЬЛіАыБчіцЎЈЈЁ3Ј©ўсЈәҙтҝӘ»оИыaЈ¬ЦрөОјУИлH2SO3ИЬТәЦБ№эБҝЈ¬H2SO3ұ»деЛ®Сх»ҜЈ¬ИЬТәөДіИ»ЖЙ«НКИҘЈ¬ЛөГчдеөҘЦКөДСх»ҜРФЗҝУЪСЗБтЛбЈ»ўтЈәПтІҪЦиўсЛщөГИЬТәЦРЦрөОјУИлH2O2ИЬТәЈ¬ИЬТәУЦұдОӘіИ»ЖЙ«Ј¬ЛөГч№эСх»ҜЗвҪ«деАлЧУСх»ҜОӘдеөҘЦКЈ¬ФтH2O2өДСх»ҜРФҙуУЪBr2Ј»ЈЁ4Ј©ІҪЦиўтЦРЈ¬ҝӘКјКұСХЙ«ОЮГчПФұд»ҜҝЙДЬКЗТтОӘІҪЦи1өДH2SO3УР№эБҝЈ¬H2O2ПИәНH2SO3·ҙУҰЈ¬»тH2O2ЕЁ¶ИРЎ»тBrЈӯУлH2O2·ҙУҰВэөИЈ»ІҪЦиўсЦРЈ¬СЗБтЛбУлдеөҘЦК·ҙУҰЈ¬АлЧУ·ҪіМКҪОӘЈәH2SO3+Br2+H2O=4H++SO42-+2Br-Ј»ІҪЦиўтЦРЦчТӘ·ўЙъ№эСх»ҜЗвәНдеАлЧУөД·ҙУҰЈ¬АлЧУ·ҪіМКҪОӘЈә H2O2+2Br-Ј«2H+ЈҪBr2Ј«2H2OЎЈ
ҝјөгЈәКөСй·Ҫ°ёөДЙијЖ
ұҫМвДС¶ИЈәА§ДС
3ЎўСЎФсМв ФЪЕЁСОЛбЦРH3AsO3УлSnCl2·ҙУҰөДАлЧУ·ҪіМ3SnCl2+12Cl-+2H3AsO3+6H+=2As+3SnCl62-+6MЈ®№ШУЪёГ·ҙУҰөДЛө·ЁЦРХэИ·өДЧйәПКЗЈЁЎЎЎЎЈ©ўЩСх»ҜјБКЗH3AsO3Ј»ўЪ»№ФӯРФЈәCl-ЈҫAsЈ»ўЫГҝЙъіЙ0.1mol?AsЈ¬·ҙУҰЦРЧӘТЖөДөзЧУөДОпЦКөДБҝОӘ0.3?molЈ®ўЬMОӘOH-Ј®
AЈ®ўЩўЫ
BЈ®ўЩўЪўЬ
CЈ®ўЪўЫўЬ
DЈ®Ц»УРўЩ
ІОҝјҙр°ёЈәУҰОӘ3SnCl2+12Cl-+2H3AsO3+6H+=2As+3SnCl62-+6MЈ®
¶ФУЪ·ҙУҰ3SnCl2+12Cl-+2H3AsO3+6H+=2As+3SnCl62-+6MЈ®
ўЩ·ҙУҰЦРЦ»УРAsФӘЛШ»ҜәПјЫҪөөНЈ¬УЙH3AsO3ЦР+3јЫҪөөНОӘ0јЫЈ¬ЛщТФH3AsO3КЗСх»ҜјБЈ¬№КўЩХэИ·Ј»
ўЪ·ҙУҰЦРЦ»УРSnФӘЛШ»ҜәПјЫЙэёЯЈ¬УЙSnCl2ЦР+2јЫЙэёЯОӘSnCl62+ЦР+4јЫЈ¬ЛщТФSnCl2КЗ»№ФӯјБЈ¬·ҙУҰЦРЦ»УРAsФӘЛШ»ҜәПјЫҪөөНЈ¬УЙH3AsO3ЦР+3јЫҪөөНОӘ0јЫЈ¬ЛщТФH3AsO3КЗСх»ҜјБЈ¬ЛщТФ»№ФӯІъОпОӘAsЈ¬»№ФӯРФSnCl2ЈҫAsЈ»·ҙУҰЦРВИФӘЛШ»ҜәПјЫОӘ·ўЙъұд»ҜЈ¬№КўЪҙнОуЈ»
ўЫ·ҙУҰЦРЦ»УРAsФӘЛШ»ҜәПјЫҪөөНЈ¬ұ»»№ФӯЈ¬УЙH3AsO3ЦР+3јЫҪөөНОӘ0јЫЈ¬ГҝЙъіЙ0.1mol?AsЈ¬·ҙУҰЦРЧӘТЖөДөзЧУөДОпЦКөДБҝОӘ0.1molЎБ3=0.3?molЈ¬№КўЫХэИ·Ј»
ўЬёщҫЭФӘЛШКШәгҝЙЦӘЈ¬MУҰОӘH2OЈ¬№КўЬҙнОуЈ®
№КўЩўЫХэИ·Ј®
№КСЎЈәAЈ®
ұҫМвҪвОцЈә
ұҫМвДС¶ИЈәјтөҘ
4ЎўСЎФсМв ҙУКҜУўЙ°ЦЖИЎІў»сөГёЯҙҝ№иөДЦчТӘ»ҜС§·ҙУҰИзПВЈә
ўЩSiO2Ј«2C Si(ҙЦ)Ј«2COЎь
Si(ҙЦ)Ј«2COЎь
ўЪSi(ҙЦ)Ј«2Cl2 SiCl4
SiCl4
ўЫSiCl4Ј«2H2 Si(ҙҝ)Ј«4HCl
Si(ҙҝ)Ј«4HCl
№ШУЪЙПКц·ҙУҰөД·ЦОцІ»ХэИ·өДКЗ
[? ]
AЈ®ўЩЎўўЫКЗЦГ»»·ҙУҰЈ¬ўЪКЗ»ҜәП·ҙУҰ
BЈ®ёЯОВПВЈ¬Ҫ№МҝУлЗвЖшөД»№ФӯРФҫщЗҝУЪ№и
CЈ®ИОТ»·ҙУҰЦРЈ¬ГҝПыәД»тЙъіЙ28?g№иЈ¬ҫщЧӘТЖ4?molөзЧУ
DЈ®ёЯОВПВҪ«КҜУўЙ°ЎўҪ№МҝЎўВИЖшЎўЗвЖш°ҙТ»¶ЁұИАэ»мәПҝЙөГёЯҙҝ№и
ІОҝјҙр°ёЈәD
ұҫМвҪвОцЈә
ұҫМвДС¶ИЈәТ»°г
5ЎўСЎФсМв ТСЦӘЈәўЩ2FeCl3+2KI=2FeCl2+2KCl+I2ўЪ2FeCl2+Cl2=2FeCl3Ј¬ФтПВБРОўБЈ»№ФӯДЬБҰУЙҙуөҪРЎөДЛіРтХэИ·өДКЗЈЁЎЎЎЎЈ©
AЈ®Fe2+ЈҫCl-ЈҫI-
BЈ®I-ЈҫFe2+ЈҫCl-
CЈ®I-ЈҫCl-ЈҫFe2+
DЈ®Cl-ЈҫI-ЈҫFe2+
ІОҝјҙр°ёЈәўЩ2FeCl3+2KI=2FeCl2+2KCl+I2ЦРЈ¬өвФӘЛШ»ҜәПјЫЙэёЯЈ¬МъФӘЛШ»ҜәПјЫҪөөНЈ¬ЛщТФВИ»ҜМъЧчСх»ҜјБЈ¬өв»ҜјШЧч»№ФӯјБЈ¬»№ФӯІъОпКЗВИ»ҜСЗМъЈ¬Фт»№ФӯРФI-ЈҫFe2+Ј¬
ўЪ2FeCl2+Cl2=2FeCl3ЦРЈ¬ВИФӘЛШ»ҜәПјЫҪөөНЈ¬МъФӘЛШ»ҜәПјЫЙэёЯЈ¬ЛщТФВИЖшКЗСх»ҜјБЈ¬ВИ»ҜСЗМъКЗ»№ФӯјБЈ¬ВИ»ҜМъКЗСх»ҜІъОпәН»№ФӯІъОпЈ¬ЛщТФ»№ФӯРФFe2+ЈҫCl-Ј¬
ФтХвИэЦЦАлЧУөД»№ФӯРФҙуРЎЛіРтКЗЈәI-ЈҫFe2+ЈҫCl-Ј¬
№КСЎBЈ®
ұҫМвҪвОцЈә
ұҫМвДС¶ИЈәТ»°г